Search Market Research Report
Pharmacovigilance Outsourcing Market Size, Share Global Analysis Report, 2026-2034

Pharmacovigilance Outsourcing Market Size, Share, Growth Analysis Report By Service (Pre-marketing Pharmacovigilance Services, Post-marketing Pharmacovigilance Services, and Others), By Provider (Contract Research Organizations, Business Process Outsourcing), By End-User (Pharmaceutical Companies, Biotechnology Companies, and Others), and By Region - Global Industry Insights, Overview, Comprehensive Analysis, Trends, Statistical Research, Market Intelligence, Historical Data and Forecast 2026-2034
Industry Insights
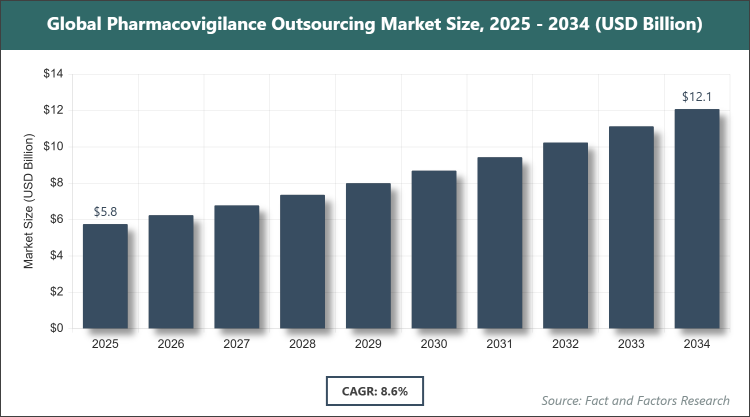
[220+ Pages Report] According to Facts & Factors, the global pharmacovigilance outsourcing market size was estimated at USD 5.75 billion in 2025 and is expected to reach USD 12.12 billion by the end of 2034. The pharmacovigilance outsourcing industry is anticipated to grow by a CAGR of 8.6% between 2026 and 2034. The pharmacovigilance outsourcing Market is driven by increasing regulatory requirements for drug safety monitoring and cost efficiencies in outsourcing operations.
 Market Overview
Market Overview
Pharmacovigilance outsourcing refers to the delegation of drug safety monitoring and adverse event reporting activities by pharmaceutical and biotechnology companies to specialized third-party service providers. This practice encompasses the collection, assessment, and analysis of data related to the safety and efficacy of medical products throughout their lifecycle, ensuring compliance with global regulatory standards and minimizing risks associated with drug usage.
 Key Insights
Key Insights
-
As per the analysis shared by our research analyst, the pharmacovigilance outsourcing market is estimated to grow annually at a CAGR of around 8.6% over the forecast period (2026-2034).
- In terms of revenue, the pharmacovigilance outsourcing market size was valued at around USD 5.75 Billion in 2025 and is expected to reach USD 12.12 Billion by 2034.
- The market is driven by rising clinical trials and stringent regulations.
- Based on the service, the post-marketing pharmacovigilance services segment dominated the market in 2025 with a share of 55% due to the higher volume of safety monitoring required after drug approval.
- Based on the provider, the contract research organizations segment dominated the market in 2025 with a share of 61% as they provide specialized expertise and end-to-end solutions.
- Based on the end-user, the pharmaceutical companies segment dominated the market in 2025 with a share of 70% owing to their extensive drug portfolios and regulatory obligations.
- Based on region, North America dominated the market in 2025 with a share of 40% due to advanced healthcare infrastructure and strict FDA regulations.
 Growth Drivers
Growth Drivers
-
 Increasing Stringent Regulatory Requirements
Increasing Stringent Regulatory Requirements
The global pharmacovigilance outsourcing market is significantly propelled by the escalating stringency of regulatory frameworks imposed by authorities such as the FDA and EMA. These regulations mandate comprehensive safety monitoring and reporting of adverse drug reactions, compelling pharmaceutical companies to adopt efficient outsourcing strategies to ensure compliance without overburdening internal resources. Outsourcing enables access to expert knowledge and advanced technologies, reducing the risk of non-compliance penalties and enhancing overall drug safety profiles.
Furthermore, the integration of real-world evidence and big data analytics in regulatory submissions has heightened the need for specialized outsourcing partners capable of handling complex data sets. This trend not only streamlines approval processes but also supports post-market surveillance, fostering market growth by allowing companies to focus on core R&D activities while maintaining high standards of patient safety.
- Rising Number of Clinical Trials and Drug Development Activities
The surge in clinical trials worldwide, driven by the increasing prevalence of chronic diseases and the demand for innovative therapies, is a key driver for pharmacovigilance outsourcing. Companies are outsourcing safety monitoring to manage the voluminous data generated during trials, ensuring timely reporting and risk assessment. This approach optimizes costs and accelerates time-to-market for new drugs.
In addition, the globalization of clinical trials necessitates multilingual and multicultural expertise, which outsourcing providers offer through their diverse operational networks. This facilitates seamless data integration across regions, improving the accuracy of safety evaluations and supporting the expansion of the market as more biopharma entities seek scalable solutions.
 Restraints
Restraints
- Data Privacy and Security Concerns
Data privacy issues pose a significant restraint to the pharmacovigilance outsourcing market, as the handling of sensitive patient information requires adherence to stringent laws like GDPR and HIPAA. Breaches can lead to severe financial and reputational damage, deterring companies from outsourcing critical functions.
Moreover, the complexity of ensuring secure data transfer and storage across international borders adds operational challenges. Providers must invest heavily in cybersecurity measures, which can increase service costs and slow down market adoption among smaller firms wary of potential vulnerabilities.
- High Costs Associated with Outsourcing Setup
Initial setup costs for outsourcing pharmacovigilance activities, including contract negotiations and system integrations, can be prohibitive for some organizations. This financial barrier limits market penetration, particularly in emerging economies where budget constraints are more pronounced.
Additionally, ongoing expenses related to quality assurance and audit compliance further strain resources. Companies may hesitate to outsource if short-term cost savings do not outweigh the long-term investments required for seamless partnership operations.
 Opportunities
Opportunities
- Adoption of AI and Machine Learning Technologies
The incorporation of AI and ML in pharmacovigilance processes presents substantial opportunities by automating adverse event detection and signal processing. Outsourcing providers leveraging these technologies can offer faster, more accurate services, attracting clients seeking efficiency gains.
This technological shift also enables predictive analytics for risk management, opening new revenue streams for providers. As adoption grows, it positions the market for innovation-driven expansion, particularly in handling large-scale data from diverse sources.
- Expansion in Emerging Markets
Emerging markets in Asia-Pacific and Latin America offer growth opportunities due to increasing pharmaceutical manufacturing and clinical research activities. Outsourcing providers can capitalize on lower operational costs and skilled labor pools in these regions to deliver cost-effective services.
Furthermore, supportive government policies and improving regulatory environments enhance market accessibility. This regional expansion allows for diversified service offerings tailored to local needs, driving overall market growth.
 Challenges
Challenges
- Lack of Standardization in Processes
The absence of uniform standards across outsourcing providers leads to inconsistencies in service quality and reporting, posing challenges for clients seeking reliable partnerships. This variability can result in compliance issues and delayed decision-making.
Efforts to harmonize processes through industry collaborations are underway, but progress is slow. Until standardization improves, it remains a hurdle affecting trust and efficiency in outsourcing arrangements.
- Skilled Workforce Shortages
A shortage of trained pharmacovigilance professionals hampers market growth, as demand outpaces supply in specialized areas like risk assessment and regulatory affairs. Outsourcing firms face recruitment challenges, impacting service delivery timelines.
Investments in training programs and partnerships with academic institutions are essential to address this gap. Without adequate talent development, the market may experience bottlenecks in scaling operations to meet rising demands.
 Report Scope
Report Scope
Report Attribute |
Details |
Market Size 2025 |
USD 5.75 Billion |
Projected Market Size in 2034 |
USD 12.12 Billion |
CAGR Growth Rate |
8.6% CAGR |
Base Year |
2025 |
Forecast Years |
2026-2034 |
Key Market Players |
IQVIA, Accenture, Cognizant, Parexel, IBM, ArisGlobal, BioClinica, Capgemini, Clinlogix, Covance, ICON, LabCorp, Syneos Health, Wipro, United BioSource, and Others. |
Key Segment |
By Service, By Provider, By End-User, and By Region |
Major Regions Covered |
North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa |
Purchase Options |
Request customized purchase options to meet your research needs. Explore purchase options |
 Market Segmentation
Market Segmentation
The pharmacovigilance outsourcing market is segmented by service, provider, end-user, and region.
Based on Service Segment, the pharmacovigilance outsourcing market is divided into pre-marketing pharmacovigilance services, post-marketing pharmacovigilance services, and others. The post-marketing pharmacovigilance services emerged as the most dominant segment, holding a significant market share due to the extensive ongoing monitoring required after drug commercialization to track long-term safety effects and comply with post-approval regulations. This segment drives the market by enabling efficient management of large-scale adverse event data, reducing internal burdens on companies, and ensuring timely regulatory submissions. The pre-marketing pharmacovigilance services stand as the second most dominant, focusing on safety assessments during clinical trials; it contributes to market growth by supporting faster drug development cycles through expert risk evaluations and data analysis, ultimately accelerating approvals and minimizing development risks.
Based on Provider Segment, the pharmacovigilance outsourcing market is divided into contract research organizations and business process outsourcing. The contract research organizations segment is the most dominant, commanding a substantial share owing to their comprehensive expertise in clinical and safety services, which allows for integrated solutions that enhance efficiency and compliance. This dominance propels the market forward by providing scalable, specialized support that helps pharmaceutical firms navigate complex regulatory landscapes and optimize resource allocation. Business process outsourcing follows as the second dominant, offering cost-effective administrative and operational handling of pharmacovigilance tasks; it aids market expansion by enabling companies to focus on core innovations while leveraging streamlined processes for data management and reporting.
Based on End-User Segment, the pharmacovigilance outsourcing market is divided into pharmaceutical companies, biotechnology companies, and others. Pharmaceutical companies represent the most dominant segment, with a large market presence driven by their vast drug pipelines and stringent safety obligations, necessitating outsourced expertise to manage voluminous data effectively. This segment fuels market growth by promoting adoption of advanced outsourcing models that improve safety outcomes and operational efficiencies. Biotechnology companies are the second most dominant, benefiting from outsourcing to handle specialized biologic safety monitoring; this supports market development by facilitating agile responses to emerging therapies and regulatory demands, enhancing innovation in the sector.
 Recent Developments
Recent Developments
- In July 2025, EVERSANA introduced EVERSANA ORCHESTRATE PV, an AI-powered solution designed to enhance pharmacovigilance efficiency through automation of case processing and signal detection, reducing manual efforts and improving accuracy in adverse event management.
- In March 2025, ArisGlobal launched three new AI-driven products LifeSphere Unify, NavaX Insights, and Advanced Compliance Docs to automate compliance and safety workflows, enabling faster data insights and better risk management for outsourcing clients.
- In June 2025, IQVIA reported strong Q1 revenue growth in its R&D Solutions, highlighting increased demand for integrated pharmacovigilance services amid a robust backlog, reflecting resilience in the outsourcing sector.
- In July 2024, Oracle updated its Argus and Safety One Intake solutions with AI features to streamline adverse event reporting and enhance real-time safety monitoring for pharmacovigilance outsourcing operations.
- In April 2025, Parexel collaborated with Palantir to integrate AI platforms into clinical data systems, aiming to improve trial efficiency and pharmacovigilance data transparency for outsourced services.
 Regional Analysis
Regional Analysis
- North America to dominate the global market
North America holds a dominant position in the pharmacovigilance outsourcing market, primarily due to its advanced healthcare infrastructure, robust regulatory environment led by the FDA, and high concentration of pharmaceutical giants. The region's emphasis on innovation and compliance drives outsourcing demand, with the United States serving as the key dominating country through its extensive clinical trial ecosystem and investment in AI-driven safety tools. This dominance fosters market stability and growth by attracting global providers and enabling seamless integration of cutting-edge technologies for enhanced drug safety monitoring.
Europe follows as a significant player, benefiting from harmonized regulations under the EMA and a strong focus on patient safety across diverse markets. Germany and the United Kingdom stand out as dominating countries, with their advanced biotech sectors and collaborative research initiatives promoting outsourcing for efficient adverse event management. The region's multicultural framework supports comprehensive data analysis, contributing to market expansion through standardized processes and increased adoption of digital solutions for pharmacovigilance.
Asia Pacific is experiencing rapid growth in the pharmacovigilance outsourcing market, driven by cost advantages, skilled workforce, and expanding clinical research activities. India and China emerge as dominating countries, leveraging their large talent pools and government support for pharma services to attract international outsourcing. This regional surge enhances global market dynamics by offering scalable, affordable solutions that accelerate drug development timelines and improve access to diverse patient data for safety assessments.
Latin America presents emerging opportunities in pharmacovigilance outsourcing, supported by improving regulatory frameworks and increasing foreign investments in healthcare. Brazil dominates the region with its growing pharma industry and focus on aligning with international standards, facilitating outsourcing for regional clinical trials. The area's development contributes to the global market by providing cost-effective alternatives and expanding the reach of safety monitoring to underrepresented populations.
The Middle East & Africa region is gradually advancing in pharmacovigilance outsourcing, aided by rising healthcare expenditures and regulatory enhancements. South Africa leads as the dominating country, with its established clinical research infrastructure attracting outsourcing partnerships. This progress supports the overall market by diversifying service offerings and addressing safety needs in emerging markets through localized expertise and international collaborations.
 Competitive Analysis
Competitive Analysis
The global pharmacovigilance outsourcing market is dominated by players:
- IQVIA
- Accenture
- Cognizant
- Parexel
- IBM
- ArisGlobal
- BioClinica
- Capgemini
- Clinlogix
- Covance
- ICON
- LabCorp
- Syneos Health
- Wipro
- United BioSource
The global pharmacovigilance outsourcing market is segmented as follows:
 By Service
By Service
- Pre-marketing Pharmacovigilance Services
- Post-marketing Pharmacovigilance Services
- Others
 By Provider
By Provider
- Contract Research Organizations
- Business Process Outsourcing
 By End-User
By End-User
- Pharmaceutical Companies
- Biotechnology Companies
- Others
 By Region
By Region
- North America
- The U.S.
- Canada
- Mexico
- Europe
- France
- The UK
- Spain
- Germany
- Italy
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- Australia
- Southeast Asia
- Rest of Asia Pacific
- The Middle East & Africa
- Saudi Arabia
- UAE
- Egypt
- Kuwait
- South Africa
- Rest of the Middle East & Africa
- Latin America
- Brazil
- Argentina
- Rest of Latin America
Table of Content
Industry Major Market Players
The global pharmacovigilance outsourcing market is dominated by players:
- IQVIA
- Accenture
- Cognizant
- Parexel
- IBM
- ArisGlobal
- BioClinica
- Capgemini
- Clinlogix
- Covance
- ICON
- LabCorp
- Syneos Health
- Wipro
- United BioSource

Copyright © 2025 - 2026, All Rights Reserved, Facts and Factors


